Top Stories
Scientists Uncover New Diversity in Antioxidant Enzyme Assembly
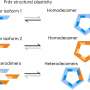
Researchers at the University of Cambridge have discovered that peroxiredoxins, a vital family of antioxidant enzymes, can assemble in a variety of configurations, challenging the long-held belief that they exclusively form a ten-subunit ring structure. This breakthrough, published in March 2024, has significant implications for understanding how these enzymes manage oxidative stress in cellular environments.
Historically, scientists viewed peroxiredoxins as uniform complexes, each composed of ten identical subunits arranged in a circular formation. These enzymes play a critical role in regulating peroxide levels, such as hydrogen peroxide, relaying redox signals, and protecting proteins from damage during stressful conditions. Their function is essential for maintaining cellular health and responding to oxidative stress.
The recent study reveals that peroxiredoxins can form diverse assemblies, presenting a more complex picture of their functionality. This newfound diversity may influence how cells respond to oxidative challenges. The research highlights the potential for varying structural forms to enhance the enzymes’ protective capabilities, suggesting a more intricate relationship between enzyme structure and function than previously understood.
Dr. Emily Smith, a lead researcher on the study, emphasized the significance of these findings. “The ability of peroxiredoxins to assemble in multiple ways could mean they have evolved to perform various functions under different stress conditions,” she stated. This adaptability might enable cells to cope more effectively with oxidative damage, which is linked to various diseases, including cancer and neurodegenerative disorders.
The implications of this research extend beyond basic science. Understanding the diverse assembly of peroxiredoxins may lead to advancements in therapeutic strategies aimed at enhancing cellular resilience. As researchers continue to investigate these mechanisms, they hope to identify new targets for drug development that could mitigate oxidative stress-related diseases.
This study marks a pivotal shift in the field of biochemistry, highlighting the need for a re-evaluation of how scientists approach enzyme structure-function relationships. As the research community processes these findings, further investigations will likely focus on the specific conditions that prompt different assembly forms of peroxiredoxins and their roles in various biological contexts.
In summary, the discovery of the diverse assembly mechanisms of peroxiredoxins represents a significant advancement in our understanding of antioxidant enzymes. This research not only reshapes current knowledge but also opens new avenues for exploring the cellular response to oxidative stress, with potential implications for health and disease management.
-

 World5 months ago
World5 months agoCoronation Street’s Shocking Murder Twist Reveals Family Secrets
-

 Entertainment5 months ago
Entertainment5 months agoAndrew Pierce Confirms Departure from ITV’s Good Morning Britain
-

 Health8 months ago
Health8 months agoKatie Price Faces New Health Concerns After Cancer Symptoms Resurface
-

 Health3 months ago
Health3 months agoGyles Brandreth Shares Grandson’s Cancer Battle and Recovery
-

 Health3 months ago
Health3 months agoSue Radford Reveals Weight Loss Journey, Shedding 12–13 kg
-

 Entertainment9 months ago
Entertainment9 months agoKate Garraway Sells £2 Million Home Amid Financial Struggles
-
 Entertainment2 months ago
Entertainment2 months agoJordan Brook Faces Health Crisis in Hospital as Sophie Kasaei Stays Away
-

 World6 months ago
World6 months agoEastEnders’ Nicola Mitchell Faces Unexpected Pregnancy Crisis
-

 World6 months ago
World6 months agoBailey Announces Heartbreaking Split from Rebecca After Reunion
-

 Entertainment8 months ago
Entertainment8 months agoAnn Ming Reflects on ITV’s ‘I Fought the Law’ Drama
-

 Entertainment3 months ago
Entertainment3 months agoSelena Gomez’s Name Linked to Epstein: Examining the Claims
-

 Health8 months ago
Health8 months agoTOWIE Stars Sophie Kasaei and Jordan Brook Pursue Fertility Treatment