Science
Revolutionary Barocaloric Technique Offers New Refrigeration Solution
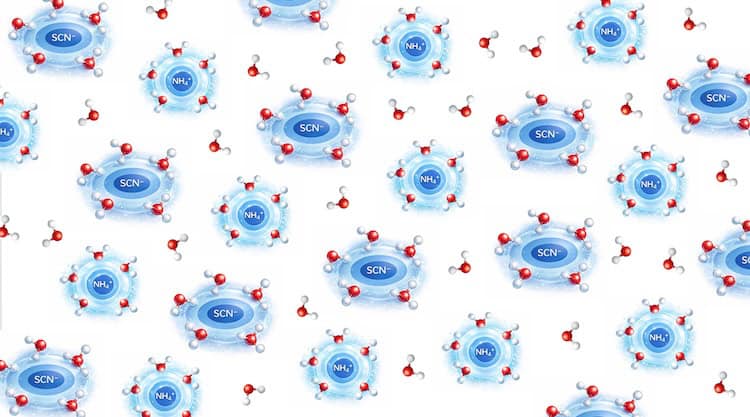
Researchers from the Institute of Metal Research of the Chinese Academy of Sciences have developed a groundbreaking cooling technique that promises to revolutionize refrigeration. This new method, based on dissolution barocaloric cooling, boasts a cooling capacity of 67 J/g and an impressive efficiency of nearly 77%. Remarkably, it can reduce the temperature of a sample by 27 K in just 20 seconds, significantly outperforming traditional barocaloric materials.
Current refrigeration technology predominantly relies on vapour-compression cooling. This century-old method involves a fluid changing phase, where a liquid refrigerant evaporates into gas, absorbing heat from its environment. A compressor then converts the gas back into a liquid, releasing heat in the process. While effective, this approach is energy-intensive and nearing the limits of efficiency as set by the Carnot limit. Moreover, many refrigerants used are toxic and contribute to environmental harm.
In recent years, researchers have turned to caloric cooling as an alternative. This approach manipulates the entropy within a material using various forces, including magnetic and electrical fields, mechanical forces, or applied pressure. Among these, barocaloric cooling has shown promise, although the majority of known barocaloric materials are solids with limited heat transfer efficiency and cooling capacity.
The innovative technique developed by Bing Li and his colleagues addresses these limitations through a fundamental thermodynamic process called endothermic dissolution. When a salt dissolves in a solvent, the energy required to break the solvent’s bonds results in cooling. The researchers utilized ammonium thiocyanate (NH4SCN), a chaotropic agent that disrupts hydrogen bonding and maximizes solubility in water. This material not only exhibits a significant enthalpy of solution but also responds effectively to applied pressures within the range of conventional hydraulic systems.
The research team’s findings, published in the journal Nature, suggest that their method could inspire further exploration into similar cooling techniques that do not depend on phase transitions. Li notes the high-temperature efficacy of aqueous NH4SCN barocaloric cooling could make it ideal for managing the thermal requirements of AI data centres, as well as potential applications in air conditioning for both domestic and industrial settings.
Despite its promising capabilities, there are challenges that must be addressed before this cooling technology can be commercialized. The corrosive nature of NH4SCN poses risks to refrigerator components, and the high pressures involved could lead to long-term damage. To tackle these issues, the researchers plan to investigate other near-saturated solutions at the atomic level, particularly their responses to pressure changes.
Li emphasized the importance of fundamental studies in optimizing the performance of these fluids as refrigerants, stating, “Such fundamental studies are vital if we are to optimize the performance of these fluids as refrigerants.” The research not only opens new avenues for environmentally friendly cooling solutions but also highlights the ongoing need for innovation in sustainable technology.
-

 World4 months ago
World4 months agoCoronation Street’s Shocking Murder Twist Reveals Family Secrets
-

 Entertainment4 months ago
Entertainment4 months agoAndrew Pierce Confirms Departure from ITV’s Good Morning Britain
-

 Health7 months ago
Health7 months agoKatie Price Faces New Health Concerns After Cancer Symptoms Resurface
-

 Health2 months ago
Health2 months agoSue Radford Reveals Weight Loss Journey, Shedding 12–13 kg
-

 Entertainment8 months ago
Entertainment8 months agoKate Garraway Sells £2 Million Home Amid Financial Struggles
-
 Entertainment3 weeks ago
Entertainment3 weeks agoJordan Brook Faces Health Crisis in Hospital as Sophie Kasaei Stays Away
-

 World5 months ago
World5 months agoEastEnders’ Nicola Mitchell Faces Unexpected Pregnancy Crisis
-

 World5 months ago
World5 months agoBailey Announces Heartbreaking Split from Rebecca After Reunion
-

 Entertainment7 months ago
Entertainment7 months agoAnn Ming Reflects on ITV’s ‘I Fought the Law’ Drama
-

 Entertainment2 months ago
Entertainment2 months agoSelena Gomez’s Name Linked to Epstein: Examining the Claims
-

 Health7 months ago
Health7 months agoTOWIE Stars Sophie Kasaei and Jordan Brook Pursue Fertility Treatment
-

 Health7 months ago
Health7 months agoFiona Phillips’ Husband Shares Heartbreaking Update on Her Health





















